What is the importance of the treat-to-target strategy in pediatric patients with IBD?
What is the importance of the treat-to-target strategy in pediatric patients with IBD?
Anne M. Griffiths, MD
Volume 2 | Issue 12
Runs 3:09
Implementation of a treat-to-target strategy is especially important in pediatric patients with inflammatory bowel disease (IBD). Pediatric patients have a particularly long remaining life span, during which intestinal complications of continuing active inflammation can occur. Moreover, unique to pediatrics is the potential, particularly in Crohn’s disease, for impairment of linear growth. This is now understood to relate to direct effects of proinflammatory cytokines released from the inflamed intestine. With traditional therapies, compromise of adult stature was formerly common. Treatments that achieve intestinal healing facilitate growth and may allow children with IBD to achieve their genetic growth potential. Pediatric IBD is highly responsive to therapy with biologics. The risk of hepatosplenic T cell lymphoma (HSTCL) with a tumour necrosis factor-α (TNF-α) inhibitor and thiopurine therapy, although rare, is an important concern, which must be addressed and discussed.
Preclinical disease and prevention
In future, identification of preclinical disease may allow interventions to target the biological processes driving disease progression and prevent clinical disease or at least modify outcomes.(1) This hope is especially important in pediatrics, as individuals diagnosed with IBD in childhood may have a more complicated course. Mitigating strategies developed for use during the preclinical phase could have the potential to affect development or aggressiveness of disease.
Growth impairment in pediatric IBD
As up to 25% of patients with CD develop disease in childhood or adolescence, the potential for growth impairment is an important complication of chronic intestinal inflammation.(2) In pediatric patients with CD, growth impairment is frequently present even before disease diagnosis and continues during the disease course, compromising stature at maturity. Such growth impairment is due primarily to proinflammatory cytokines released from the inflamed intestine. These cytokines not only reduce appetite, leading to poor intake and nutritional insufficiency, but also interfere directly with the actions of insulin growth factor-1 (IGF-1) at the epiphyses of long bones. Chronic corticosteroid use may worsen the situation. Effective treatment with TNF-α inhibitors to achieve intestinal healing has improved the growth of young patients, as long as treatment is initiated early enough before or during early puberty. Early diagnosis, effective control of inflammation, and adequate nutrition can prevent or ameliorate growth impairment.
Biologics in pediatric IBD
The benefits of early use of TNF-α inhibitor therapy, including efficacy and reduction or avoidance of growth impairment, are being increasingly recognized, especially in children with a high risk of aggressive disease.
Infliximab pharmacokinetics
Efficacy of biologics in adults with IBD can be extrapolated to children, but specifically pediatric pharmacokinetic studies are needed to ascertain optimal dosing. The pharmacokinetics of infliximab have been studied in pediatric patients with ulcerative colitis (UC).(3) Infliximab pharmacokinetics, the exposure-response relationship in pediatric patients with UC, and the relation between infliximab serum levels and efficacy were comparable to observations in adults with UC, supporting the use of adult induction and maintenance dosing in children. Canadian pediatric practice, however, has moved to monitoring drug levels prior to maintenance therapy to optimize regimens in individual patients. Multicentre pharmacokinetic studies are underway to determine the dosing required in acute severe colitis, where drug is cleared rapidly.
Early initiation of infliximab
An analysis of data from the RISK study in children with CD compared the effectiveness of early (≤3 months after diagnosis) treatment with a TNF-α inhibitor with immunomodulator treatment in achieving clinical remission and facilitating growth.(4) Significantly more (P=.0017) children were in remission at 1 year with early TNF-α inhibitor treatment (85.3%) than early immunomodulator therapy (60.3%). A greater mean height z-score increase from baseline was seen in the early TNF-α inhibitor treatment group.
A single-centre retrospective cohort study of the use of infliximab ± an immunomodulator to treat pediatric CD (N=195) evaluated clinical response, mucosal healing, linear growth and durability of response.(5) Results included an 81% complete clinical remission rate following 3-dose induction. Treatment optimization (per kg dose increase and/or interval shortening) was performed in 35% of patients during the first year, based on return of symptoms prior to infusions. Loss of responsiveness occurred at an annual rate of 2 to 6% (25% over 5 years), almost always due to the development of antidrug antibodies (ADAs). Concurrent immunomodulator use (either azathioprine or methotrexate) resulted in lower rates of ADA development, and significantly improved durability of response (P=.014). Use of methotrexate, rather than a thiopurine, may be preferred to address concerns about HSTCL. There was significant improvement (P<.001) in mean height z-score after 1 and 2 years of follow-up, and normalization of the mean height z-score after 3 years in patients at Tanner stage 1 or 2 of pubertal development when infliximab was initiated. A 73% rate of mucosal healing was observed in those undergoing repeat colonoscopy when in clinical remission.
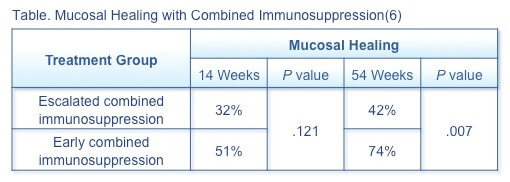
A prospective observational study compared the efficacy at 14 and 54 weeks of escalated combined immunosuppression (n=28) and early combined immunosuppression (n=48) with infliximab in achieving mucosal healing in pediatric patients with moderate-to-severe CD.(6) Patients in the escalated combined immunosuppression group had a significantly (P<.001) longer diagnosis-to-treatment interval (median 8.1 months) than in the early combined immunosuppression group (.7 months). Mucosal healing was more effectively achieved with early combined immunosuppression (Table) than escalated therapy.
Treat to target
The Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) program examined potential targets and the evidence supporting them and developed consensus recommendations for treatment targets for UC and CD.(7)
Ulcerative colitis
The program agreed that the target for UC was the following:(7)
- Clinical and patient-reported remission (PRO), defined as resolution of rectal bleeding and diarrhea or altered bowel habit (assessed at least every 3 months until resolution, then every 6 to 12 months), plus
- Endoscopic remission, defined as a Mayo endoscopic subscore of 0 or 1 (assessed 3 to 6 months after starting therapy in symptomatic patients and then at least every 3 months during active disease).
STRIDE considered histologic remission to be an adjunctive target, as insufficient evidence exists to recommend histologic remission as a target in clinical practice.
Crohn’s disease
The agreed target for CD was the following:(7)
- Clinical and PRO, defined as resolution of abdominal pain and diarrhea or altered bowel habit (assessed at least every 3 months until resolution, then every 6 to 12 months), plus
- Endoscopic remission, defined as resolution of ulceration at ileocolonoscopy, or resolution of inflammation on cross-sectional imaging, in patients who could not be assessed adequately by ileocolonoscopy (assessed at least every 6 to 9 months during active disease).
STRIDE considered biomarker remission, that is, normalization of C-reactive protein and fecal calprotectin to be an adjunctive target, as insufficient evidence exists to recommend treatment optimization based on biomarkers alone.
Conclusion
Early initiation of combined immunosuppression with TNF-α inhibitor therapy in pediatric patients with CD can reduce complications, including growth impairment. The treat-to-target strategy is especially important in pediatrics, and therapeutic drug monitoring is a useful tool to support treatment optimization.
References
- Torres J, Burisch J, Riddle M, et al. Preclinical disease and preventive strategies in IBD: perspectives, challenges and opportunities. Gut. 2016;65(7):1061-9.
- Walters TD, Griffiths AM. Mechanisms of growth impairment in pediatric Crohn’s disease. Nat Rev Gastroenterol Hepatol. 2009;6:513–23.
- Adedokun OJ, Xu Z, Padgett L, et al. Pharmacokinetics of infliximab in children with moderate-to-severe ulcerative colitis: results from a randomized, multicenter, open-label, phase 3 study. Inflamm Bowel Dis. 2013;19:2753–62.
- Walters TD, Kim MO, Denson LA, et al. Increased effectiveness of early therapy with anti-tumor necrosis factor-alpha vs an immunomodulator in children with Crohn’s disease. Gastroenterology. 2014;146:383–91.
- Church PC, Guan J, Walters TD, et al. Infliximab maintains durable response and facilitates catch-up growth in luminal pediatric Crohn’s disease. Inflamm Bowel Dis. 2014;20:1177–86.
- Kang B, Choi SY, Kim HS, et al. Mucosal healing in paediatric patients with moderate-to-severe luminal Crohn’s disease under combined immunosuppression: escalation versus early treatment. J Crohns Colitis. 2016 Apr 19. pii: jjw086. [Epub ahead of print]
Special Edition IBD Dialogue 2016 Volume 02: Treat-to-Target in IBD is made possible by an unrestricted educational grant from…
![]()
