Modern Use of TDM Across Agents
Modern Use of TDM Across Agents
May 11, 2021
Introduction
Therapeutic drug monitoring (TDM) is a tool that can be used to optimize biologic therapies in patients with inflammatory bowel disease (IBD). Not all patients require TDM, but in some it can help to reach a therapeutic target. For new biologics that have come onto the market and which may be less immunogenic, the focus has shifted from ruling out immunogenicity to confirming adequate drug exposure. Patients at risk of accelerated drug clearance—and therefore subtherapeutic drug exposure—can be identified prior to initiation and soon after initiation of therapy.
The modern use of TDM across agents helps to identify those patients who have a lower likelihood of achieving a therapeutic target due to immune- or non-immune-mediated pharmacokinetic (PK) failure. Rapid lateral-flow based assays, together with dosing dashboards, now allow for precision dosing at point of care. This brief review discusses the state of the art in TDM and implications for the future management of IBD.
Why TDM? Inter- and intra-individual Variability in PK and the PK-PD Relationship
Patients with IBD are a heterogeneous group in many respects. In the context of TDM, the critical area of heterogeneity is the inter-individual PK variability. Indeed, there is also considerable intra-patient variability in PK.
A number of variables influence variability in PK. These include differences in dosing (e.g., interval, route of administration); and patient-related factors (e.g., gender, body weight, albumin level, inflammatory burden, immunogenicity, concomitant medications and genetic variation).(1)
As an example of a dosing difference, vedolizumab has a different exposure profile when administered subcutaneously vs. intravenously.(2) Among the patient-related factors, inflammatory burden is a particularly important variable; all of the anti-TNF agents, as well as vedolizumab and ustekinumb, have been associated with increased clearance in the setting of active inflammation (as measured by c-reactive protein [CRP], fecal calprotectin or endoscopy).(3-10)
Immunogenicity is another critical factor, associated with increased clearance of anti-TNF agents and vedolizumab.(5,6,10–16) There are a number of product-related factors that can contribute to immunogenicity, including production-related factors, handling and storage.(17) Among the patient-related factors, those that are particularly important for clinicians are: route of administration, genetic factors, concomitant diseases and/or medications and history of prior anti-drug antibodies (ADAs).(17) Whether an agent is administered subcutaneously or intravenously has been shown to affect rates of immunogenicity with some biologics used for IBD. A randomized, controlled trial comparing intravenous (IV) and subcutaneous (SC) formulations of a biosimilar infliximab (CT-P13) showed that the proportion of patients with neutralizing antibodies (NAb) was markedly higher in the IV group.(18,19)
With respect to genetic factors, a genome-wide association study published in 2020 identified the presence of the HLA-DQA1*05 allele (carried by approximately 40% of Europeans) as a significant risk factor for the development of ADAs to infliximab or adalimumab among patients with Crohn’s disease (CD): overall hazard ratio 1.9; 95% CI 1.60–2.25 vs. non-carriers.(20) The study further characterized the increased rates of immunogenicity among those carrying the allele as being higher among those treated with infliximab compared to adalimumab, and higher among those who were on monotherapy compared to those receiving the anti-TNF agent in combination with an immunomodulator.(20)
Also, with respect to anti-TNF therapy, recent research has shown that for patients switching from one anti-TNF therapy to another, those with a prior history of antibodies to the first agent have an increased risk of developing ADAs to the second.(21) This was found to be true whether the sequence was an infliximab-to-adalimumab switch or vice-versa.
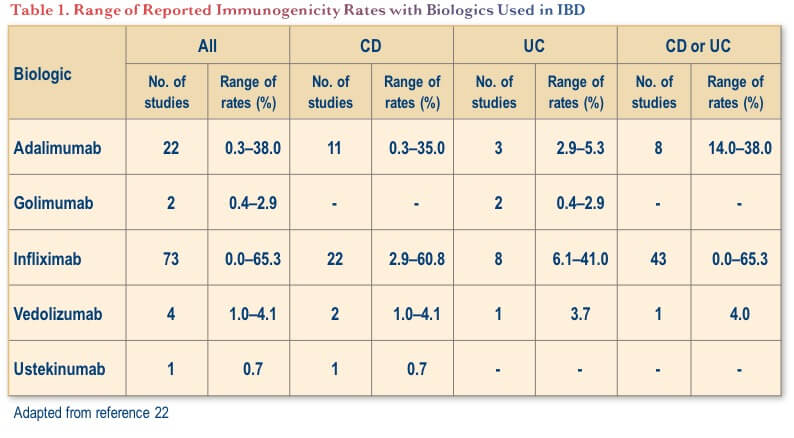
Notably, while immunogenicity is a key consideration with anti-TNF therapies, it appears to be less of a concern with newer biologics with other mechanisms of action used for IBD (i.e., vedolizumab and ustekinumab).(22) As shown in Table 1, the range of rates of ADA formation to these other biologics has been low in both CD and ulcerative colitis (UC), albeit from a smaller pool of studies. Nevertheless, rates of immunogenicity are difficult to compare across agents because of analytical confounders, dosing regimen and concomitant medication.
Who Gets TDM, and When? Patient Selection
Deciding when to order TDM is a key consideration in optimizing biologic treatment for individuals with IBD. There is a large and growing body of research that has investigated the utility of TDM at secondary loss of response (i.e., reactive TDM), or routinely at specific time points in therapy (i.e., proactive TDM), including comparisons of the two approaches. The following section provides an overview of key studies in this field.
Reactive TDM
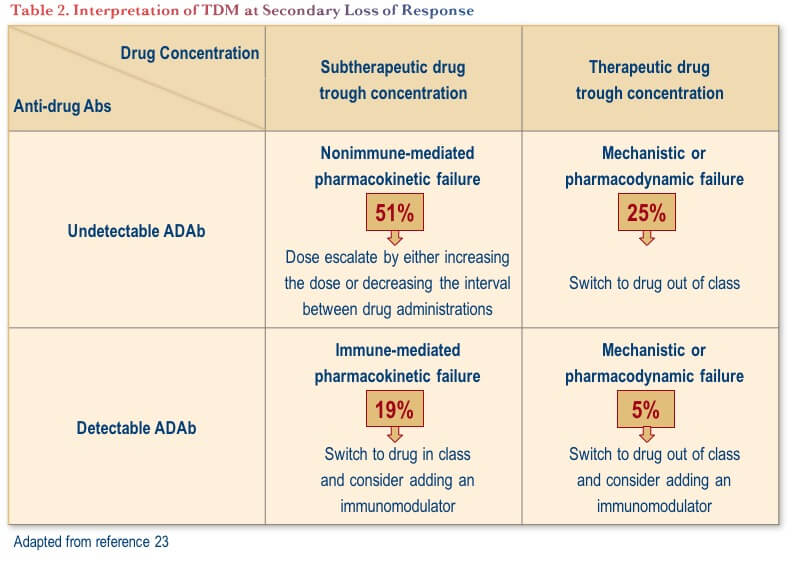
TDM performed in reaction to secondary loss of response provides important information to direct the subsequent approach to therapy. A technical review conducted by the American Gastroenterological Association Institute proposed evidence-based interventions based on reactive TDM, as outlined in Table 2.(23) From a practical perspective, the table shows that one should look at trough drug concentrations first; if they are optimal, then ADAs are probably inconsequential. If the trough drug level is subtherapeutic, it is important to understand what is causing accelerated drug clearance and thus confirming the presence or absence of ADAs is an important consideration in this scenario.
The use of reactive TDM for secondary anti-TNF failure is supported by prospective clinical trial evidence showing that basing therapeutic decisions on reactive TDM is significantly more cost-effective than increasing the dose of the anti-TNF agent.(24)
Proactive TDM
There are now several published prospective studies that have investigated the utility of proactive TDM for therapeutic decision making with TNF inhibitors in IBD. The results have been somewhat equivocal, as documented below.
Infliximab
The prospective TAXIT trial sought to determine whether infliximab dosing based on TDM was effective in improving remission rate during an initial optimization phase of treatment for CD or UC (n=148) and whether it was superior to clinically-based decision making during the randomized maintenance (n=251).(25) During optimization, TDM-based dose escalation led to better clinical and biochemical outcomes, including an increase in remission rate among CD patients from 65.1% prior to dose escalation to 88.4% after escalation. Additionally, TDM-based dose de-escalation did not affect disease activity and reduced drug costs by 28%.(25) During the randomized maintenance phase, there was no significant difference between TDM- or clinically-based decision making with respect to maintenance of combined clinical and biochemical remission, one year after the initial optimization phase. However, significantly fewer patients in the TDM group required rescue therapy for flares (7% vs. 17%, p=0.018) and a significantly greater proportion of patients in the TDM group stayed in the infliximab target range (3–7 mcg/mL) (74% vs. 57%; p=0.001).(25)
The TAILORIX study was a prospective study that included 122 patients with CD treated with infliximab. The subjects were randomized to receive dose increases based on clinical assessment alone or based on clinical assessment, biomarker analysis (CRP and fecal calprotectin) and TDM based on trough concentration.(26) In this study, for the primary endpoint of sustained corticosteroid-free clinical remission (from weeks 22 through 54, with no ulcers at week 54), there was no significant difference between groups.
The PRECISION study was a prospective, randomized study involving 80 patients with CD or UC in clinical remission on infliximab maintenance therapy. Subjects were randomized to a “precision” group, whose infliximab dosing was guided based on a target trough drug level of 3 mcg/mL; or a control group, where infliximab treatment was continued without dose adaptations. In this study, there was a significant difference in sustained clinical remission rates at one year: 28/32 (88%) of patients in the precision group vs. 25/39 (64%) in the control group (p=.017).(27)
Adalimumab
With adalimumab, there has been one prospective study that compared proactive and reactive approaches to adalimumab dosing during maintenance.(28) The trial was conducted among 78 pediatric subjects (aged 6–18 years) with luminal CD. Patients were randomized to receive proactive monitoring of trough concentrations at weeks 4 and 8 and then every eight weeks, or reactive monitoring (physicians were informed of trough concentrations after loss of response). The target in both groups was an adalimumab trough concentration of 5 mcg/mL. In this study, the proactive strategy was associated with significantly higher rates of sustained corticosteroid-free clinical remission at all visits from week 8 through week 72 (82% of the proactive group vs. 48% in the reactive group, p=0.002).(28)
TDM has also been used in other recent studies investigating the optimal dose and frequency of adalimumab in IBD. The SERENE studies (SERENE-UC and SERENE-CD) were conducted to determine the impact of higher doses of adalimumab on the likelihood of remission in induction and maintenance therapy.(29-32)
While TDM conducted throughout the induction studies showed that there were significant differences in adalimumab trough concentration, which peaked at week 4 in the higher-dose groups, these were not associated with any significant differences in efficacy endpoints.(29,30)
In the maintenance arm of the SERENE-UC study, clinical remission at Week 52 was found to be numerically but not statistically higher in patients receiving adalimumab 40 mg every week vs. 40 mg every other week during maintenance.(31) Similarly, in the SERENE-CD study, there were no significant efficacy differences between higher or standard maintenance doses.(32) In an additional analysis of the SERENE-CD maintenance study that compared outcomes between subjects who had their adalimumab doses adjusted based on clinical symptoms and biomarkers, or based on symptoms, biomarkers, and adalimumab serum levels, there was no additional benefit associated with the proactive TDM strategy.(32)
Vedolizumab
To date, no trials conducted with vedolizumab have evaluated proactive or reactive TDM. However, evidence from the GEMINI clinical trial program suggests that there is an apparent exposure-response relationship.(33) In a retrospective cohort study of 179 consecutive patients (66 with UC and 113 with CD) in Belgium, vedolizumab trough concentration thresholds associated with better outcomes were: greater than 30 mcg/mL at Week 2; greater than 24 mcg/mL at Week 6 and greater than 14 mcg/mL during maintenance.(34) Real-world evidence from a pooled multicentre observational cohort analysis of clinical and modelled pharmacological data produced similar results: the predicted vedolizumab exposure thresholds for deep remission were 30.7 mcg/mL at Week 6, 32.1 mcg/mL at week 10 and 21.1 mcg/mL at week 14.(35)
In the VISIBLE trial, which evaluated vedolizumab SC or IV formulations for maintenance following IV induction, investigators included an analysis of clinical remission by quartile of trough vedolizumab concentration.(36) In both the SC and IV arms, the proportion of patients who achieved clinical remission at week 52 increased with increasing vedolizumab exposure.
Ustekinumab
With ustekinumab, an apparent exposure-response relationship was described in the UNITI clinical trial program.(10) In a small, prospective, real-world analysis of 62 patients with CD, thresholds associated with better outcomes were >0.8–1.35 mcg/mL for maintenance of clinical remission; and >4.5 μg/mL for maintenance of endoscopic response.(37) In that study, trough levels below 0.5 mcg/mL were associated with worse outcomes during ustekinumab maintenance. Further evidence supporting increased efficacy with higher ustekinumab trough concentrations comes from the STARDUST treat-to-target study in CD, where increasing quartiles of ustekinumab trough concentration were associated with improved endoscopic response and remission rates.(38)
Conclusions
Precision medicine in IBD may involve a combination of genetic testing, drug selection based on product- or patient-specific factors and TDM. New formulations may improve the pharmacokinetics of established agents (e.g., infliximab, vedolizumab). Besides trough concentrations, other exposure measures may be important (e.g., clearance, average concentration). Reactive TDM will continue to be an important tool at time of loss of response while pharmacokinetics coupled with the use of TDM can be used to identify patients most likely to benefit from proactive TDM.
Clinical Case
April is a 24-year-old female working as a laboratory technician. She has an eight-year history of left-sided UC, which had been previously controlled with 4.8 g of mesalamine. After a disease flare two years ago she required a weaning course of oral corticosteroids. She had another flare six months later, but declined a further trial of corticosteroids due to concerns of weight gain and mood changes. She was induced with vedolizumab one year ago and subsequently maintained on 300 mg, administered intravenously (IV) every eight weeks.
For the last four months she has had urgency and bloody diarrhea with four to six bowel movements (BMs) per day, abdominal cramps, and a sensation of incomplete evacuation. Her investigations revealed:
- WBC: 8.0 x 109/L
- Hb: 102 g/L
- CRP: 14 mg/L
- Albumin: 36 g/L
- Fecal calprotectin: 2100 mcg/g
- Stool C and S negative
- C. difficile negative
- Mayo 2 pancolitis on colonoscopy.
Commentary
In a 2019 consensus paper on TDM for biologic agents, the recommendations for vedolizumab included the following: “It is appropriate to order drug/antibody concentration testing for vedolizumab in patients with confirmed secondary loss of response.”(39) This fits the current clinical situation.
A systematic review and meta-analysis published in 2019 evaluated the potential role of TDM for vedolizumab.(40) The analysis included five cohort studies involving a total of 558 patients (42% with UC) reporting the association between vedolizumab trough concentration and clinical outcomes in patients with IBD. Vedolizumab trough concentrations were consistently higher in patients achieving clinical remission vs. no clinical remission (median 14.3 mcg/mL vs. 10.5 mcg/mL) or endoscopic remission vs. no endoscopic remission (median 13.0 mcg/mL vs 9.7 mcg/mL). In patients with UC, week 6 vedolizumab trough concentrations ≥18.5–20.8 mcg/mL, and maintenance trough concentrations ≥9.0–12.6 mcg/mL were associated with favourable clinical outcomes.
The subsequent course of action would be informed by the findings from the TDM. For example:(40)
- If the concentration is low, but close to therapeutic range, one might consider dose optimizing the vedolizumab (e.g., shortening the dosing interval between infusions from every eight to every four weeks) to recapture the remission she previously had with this agent.
- If the concentration is subtherapeutic and there are high-titer antibodies to vedolizumab, one would not expect that dose optimization would be effective and a switch to another therapy is warranted.
Case Evolution
You decide to check a vedolizumab level which returns at 15.5 mcg/mL. After discussion with the patient, you decide to dose escalate to vedolizumab 300 mg infusions every four weeks for four months, then re-evaluate accordingly. Unfortunately, her symptoms progress and she requests to change therapies.
Commentary
At this point, there is likely no point in checking her vedolizumab levels, as further dose escalation with vedolizumab is not feasible. Assuming that her adherence with vedolizumab has been good, the request to change therapies is more than reasonable. The Canadian guidelines for the management of UC suggest that for patients who fail therapy with vedolizumab, they be switched to an anti-TNF biologic.(41) Since the publication of these guidelines (2015), however, there have been additional potential therapies added to the treatment landscape, namely ustekinumab (an anti-IL-12/23 biologic) and tofacitinib, an oral Janus kinase inhibitor small molecule.(42) There is limited evidence to suggest that one type of subsequent therapy can be expected to be better than others following failure of vedolizumab: Any of the above options can be considered in this case and patient preference should play the key role.
Case Evolution
April requests to switch to ustekinumab as she would like a medication she can self-inject. Despite your counselling regarding the relative safety of all biologic agents, she is unduly worried about the risks of anti-TNF agents. You arrange an IV induction of ustekinumab, then maintenance with SC dosing every eight weeks and she has an initial clinical response. Before her third SC dose, she requests that you check her ‘drug level’. Fortunately, she has maintained clinical remission.
Commentary
At this point in therapy, there is no clinical rationale for performing TDM. The 2019 consensus paper on TDM gave the following guidance for TDM with ustekinumab:(39)
- It is appropriate to order drug/antibody concentration testing for ustekinumab in non-responders at the end of induction (at 8 weeks); and
- It is appropriate to order drug/antibody concentration testing for ustekinumab in patients with confirmed secondary loss of response.
Neither of these fits the current scenario, and proactive TDM for ustekinumab is not currently supported by guidelines or evidence.(10,37,39,43–45)
With respect to other biologics (i.e., anti-TNF agents), there is substantially more evidence available to make decisions regarding proactive TDM. The 2019 consensus paper on TDM made the following recommendation:(39)
- It is appropriate to order drug/antibody concentration testing in responders at the end of induction for all anti-TNFs; and
- It is appropriate to order drug/antibody concentration testing at least once during maintenance for patients on all anti-TNFs.
Case Evolution
You decline April’s request to check her drug level and she enjoys excellent quality of life and continues to work at the laboratory. Unfortunately, at her 18-month follow-up visit, she begins experiencing a relapse, with a recurrence of four to six bloody BMs per day, CRP 15 mg/L and fecal calprotectin 1800 mcg/g.
Commentary
As mentioned above, the 2019 consensus paper on TDM states that it is appropriate to order drug/antibody concentration testing for ustekinumab in patients with confirmed secondary loss of response,(39) which fits the current situation. If the concentration is found to be low, dose optimization can be considered.
Different studies have proposed different thresholds for a therapeutic concentration of ustekinumab during maintenance for IBD, from above 1 mcg/mL to above 4.5 mcg/mL, all while acknowledging that the evidence supporting these thresholds is minimal.(10,37,44,45)
With respect to the target ranges, studies investigating proactive or reactive TDM for anti-TNF agents have produced some potentially important differences in recommended thresholds and ranges. Based on the available data for proactive TDM, for example (e.g., the prospective TAXIT, TAILORIX and PAILOT studies)(25,26,28), proposed ranges for infliximab and adalimumab are as follows:(46,47)
- Infliximab induction (week 10):
- >25 mcg/mL: no adjustment
- 20–25 mcg/mL: only frequency adjustment
- <20 mcg/mL: dose and frequency adjustment
- Infliximab maintenance (6 months):
- >7 mcg/mL: No adjustment / consider adjustment for mucosal healing
- 3–7 mcg/mL only frequency adjustment
- <3 mcg/mL: dose and frequency adjustment
- Adalimumab induction (week 4):
- <12 mcg/mL: frequency adjustment
- Adalimumab maintenance (6 months):
- <5 mcg/mL: frequency adjustment
For reactive TDM, differing thresholds have been suggested. For example, the American Gastroenterological Association’s recommendations suggest target trough concentrations for reactive TDM among patients with active IBD during maintenance:(48)
- Infliximab ≥5 mcg/mL
- Adalimumab ≥7.5 mcg/mL
Case Evolution
A serum trough level of ustekinumab returns at 1.4 mcg/mL. She declines dose optimization, stating that higher doses will increase her risk of COVID as the pandemic is now in full force. You counsel her at length about the need for ongoing disease control but are informed by your patient support coordinator six months later that April has not picked up any injections since you last saw her. You try calling her and send out a fecal calprotectin kit, which returns at >5400 mcg/g. Two weeks later, she attends the emergency department with 10 bloody BMs per day, three nocturnal BMs per night, CRP of 22 mg/L and albumin of 26 g/L.
Commentary
The ustekinumab level may indeed be subtherapeutic, based on the range of proposed thresholds in the literature (from above 1 mcg/mL to above 4.5 mcg/mL).(10,37,44,45) However, given the severity of her symptoms (and also given that she will not consent to dose optimization), a switch of therapy is in order. A course of intravenous steroids should be administered in hospital.(49) If infliximab is administered in this acute setting, the severity of her current symptoms suggests that dose optimization at 10 mg/kg should be considered.(50)
Once stabilized and back in the community, TDM and adjustment of dose should be considered.
Case Concludes
Fortunately, after receiving intravenous steroids and 10 mg/kg of infliximab at day 0 and 7, April does well, and is discharged on oral prednisone and has a plan for outpatient infliximab infusions.
References
-
Lefevre PLC, Shackelton LM, Vande Casteele N. Factors Influencing Drug Disposition of Monoclonal Antibodies in Inflammatory Bowel Disease: Implications for Personalized Medicine. BioDrugs. 2019;33(5):453–68.
-
Rosario M, Polhamus D, Chen C, et al. A vedolizumab population pharmacokinetic model including intravenous and subcutaneous formulations for patients with ulcerative Colitis [Abstract P490]. J Crohns Colitis. 2019;13(Suppl 1):357.
-
Hemperly A, Vande Casteele N. Clinical pharmacokinetics and pharmacodynamics of infliximab in the treatment of inflammatory bowel disease. Clin Pharmacokinet. 2018;57:929–42.
-
Sharma S, Eckert D, Hyams JS, et al. Pharmacokinetics and exposure-efficacy relationship of adalimumab in pediatric patients with moderate to severe Crohn’s disease: results from a randomized, multicenter, phase-3 study. Inflamm Bowel Dis. 2015;21:783–92.
-
Wade JR, Parker G, Kosutic G, et al. Population pharmacokinetic analysis of certolizumab pegol in patients with Crohn’s disease. J Clin Pharmacol. 2015;55:866–74.
-
Vande Casteele N, Mould DR, Coarse J, et al. Accounting for pharmacokinetic variability of certolizumab pegol in patients with Crohn’s disease. Clin Pharmacokinet. 2017;56:1513–23.
-
Adedokun OJ, Xu Z, Marano CW, et al. Pharmacokinetics and exposure-response relationship of golimumab in patients with moderately-to-severely active ulcerative colitis: results from phase 2/3 PURSUIT induction and maintenance studies. J Crohns Colitis. 2017;11:35–46.
-
Ward MG, Sparrow MP, Roblin X. Therapeutic drug monitoring of vedolizumab in inflammatory bowel disease: current data and future directions. Ther Adv Gastroenterol. 2018;11:1–10.
-
Dreesen E, Verstockt B, Bian S, et al. Evidence to support monitoring of vedolizumab trough concentrations in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol. 2018;16:1937–46.
-
Adedokun OJ, Xu Z, Gasink C, et al. Pharmacokinetics and exposure response relationships of ustekinumab in patients with Crohn’s disease. Gastroenterology. 2018;154:1660–71.
-
Fasanmade AA, Adedokun OJ, Blank M, et al. Pharmacokinetic properties of infliximab in children and adults with Crohn’s disease: a retrospective analysis of data from 2 phase III clinical trials. Clin Ther. 2011;33:946–64.
-
Fasanmade AA, Adedokun OJ, Ford J, et al. Population pharmacokinetic analysis of infliximab in patients with ulcerative colitis. Eur J Clin Pharmacol. 2009;65:1211–28.
-
Berends SE, Strik AS, Van Selm JC, et al. Explaining interpatient variability in adalimumab pharmacokinetics in patients with Crohn’s disease. Ther Drug Monit. 2018;40:202–11.
-
Ternant D, Karmiris K, Vermeire S, et al. Pharmacokinetics of adalimumab in Crohn’s disease. Eur J Clin Pharmacol. 2015;71:1155–7.
-
Verstockt B, Moors G, Bian S, et al. Influence of early adalimumab serum levels on immunogenicity and long-term outcome of anti-TNF naïve Crohn’s disease patients: the usefulness of rapid testing. Aliment Pharmacol Ther. 2018;48:731–9.
-
Xu Y, Adedokun OJ, Chan D, et al. Population pharmacokinetics and exposure-response modeling analyses of golimumab in children with moderately to severely active ulcerative colitis. J Clin Pharmacol. 2018;59:590–604.
-
Schellekens H. Bioequivalence and the immunogenicity of biopharmaceuticals. Nat Rev Drug Discov. 2002;1(6):457–62.
-
Schreiber S, Leszczyszyn J, Dudkowiak R, et al. Noninferiority of novel subcutaneous infliximab (CT-P13) in patients with active Crohn’s disease and ulcerative colitis: Week 30 results from a multicentre, randomised controlled pivotal trial [abstract LB02]. United Eur Gastroenterol J. 2019;7(10):1412–13.
-
Schreiber S, Ben-Horin S, Leszczyszyn J, et al. Randomized Controlled Trial: Subcutaneous versus Intravenous Infliximab CT-P13 Maintenance in Inflammatory Bowel Disease. Gastroenterology. 2021;Mar 4 [Epub ahead of print].
-
Sazonovs A, Kennedy NA, Moutsianas L, et al. HLA-DQA1*05 Carriage Associated With Development of Anti-Drug Antibodies to Infliximab and Adalimumab in Patients With Crohn’s Disease. 2020;158(1):189–99.
-
Vande Casteele N, et al. Unpublished data, 2021.
-
Vermeire S, Gils A, Accossato P, et al. Immunogenicity of biologics in inflammatory bowel disease. Therap Adv Gastroenterol. 2018;11:1756283X17750355.
-
Vande Casteele N, Herfarth H, Katz J, et al. American Gastroenterological Association Institute Technical Review on the Role of Therapeutic Drug Monitoring in the Management of Inflammatory Bowel Diseases. Gastroenterology. 2017;153(3):835–57.
-
Steenholdt C, Brynskov J, Thomsen OØ, et al. Individualised therapy is more cost-effective than dose intensification in patients with Crohn’s disease who lose response to anti-TNF treatment: a randomised, controlled trial. Gut. 2014;63(6):919–27.
-
Vande Casteele N, Ferrante M, Van Assche G, et al. Trough concentrations of infliximab guide dosing for patients with inflammatory bowel disease. Gastroenterology. 2015;148(7):1320–9.
-
D’Haens G, Vermeire S, Lambrecht G, et al. Increasing Infliximab Dose Based on Symptoms, Biomarkers, and Serum Drug Concentrations Does Not Increase Clinical, Endoscopic, and Corticosteroid-Free Remission in Patients With Active Luminal Crohn’s Disease. Gastroenterology. 2018;154(5):1343–51.
-
Strik AS, Löwenberg M, Mould DR, et al. Efficacy of dashboard driven dosing of infliximab in inflammatory bowel disease patients; a randomized controlled trial. Scand J Gastroenterol. 2021;56(2):145–54.
-
Assa A, Matar M, Turner D, et al. Proactive Monitoring of Adalimumab Trough Concentration Associated With Increased Clinical Remission in Children With Crohn’s Disease Compared With Reactive Monitoring. Gastroenterology. 2019;157(4):985–96.
-
Panés J, Colombel JF, D’Haens G, et al. High versus standard adalimumab induction dosing regimens in patients with moderately to severely active ulcerative colitis: results from the SERENE-UC induction study [UEG Week abstract OP216]. United European Gastroenterol J. 2019;7(8 suppl).
-
D’Haens G, Sandborn WJ, Loftus EV, et al. High versus standard adalimumab induction dosing regimens in patients with moderately to severely active Crohn’s disease: results from the SERENE-CD induction study [UEG Week abstract LB27]. United European Gastroenterol J. 2019;7(8 suppl).
-
Colombel JF, Panés J, D’Haens G, et al. Higher vs. standard adalimumab maintenance regimens in patients with moderately to severely active ulcerative colitis: Results from the SERENE-UC maintenance study [ECCO abstract OP01]. J Crohn’s Colitis. 2020;S001.
-
Danese S, Sandborn WJ, Loftus EV Jr., et al. Clinically adjusted versus therapeutic drug monitoring dosing regimens with adalimumab patients with moderately to severely active Crohn’s disease: Results from the SERENE-CD maintenance study [UEG Week abstract OP043]. United European Gastroenterol J. 2020;8(Suppl):34.
-
Feagan BG, Rutgeerts P, Sands BE, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013;369(8):699–710.
-
Dreesen E, Verstockt B, Bian S, et al. Evidence to Support Monitoring of Vedolizumab Trough Concentrations in Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2018;16(12):1937–46.
-
Vande Casteele N, Sandborn W, Feagan BG, et al. Real-world exposure-response relationship of vedolizumab in inflammatory bowel disease: A pooled multicentre observational cohort analysis of clinical and modeled pharmacological data [UEG Week abstract P0503]. United European Gastroenterol J. 2020;8(Suppl):34.
-
Sandborn WJ, Baert F, Danese S, et al. Efficacy and Safety of Vedolizumab Subcutaneous Formulation in a Randomized Trial of Patients With Ulcerative Colitis. Gastroenterology. 2020;158(3):562–72.
-
Battat R, Kopylov U, Bessissow T, et al. Association Between Ustekinumab Trough Concentrations and Clinical, Biomarker, and Endoscopic Outcomes in Patients With Crohn’s Disease. Clin Gastroenterol Hepatol. 2017;15(9):1427–34.
-
Danese S, Vermeire S, D’Haens GR, et al. Pharmacokinetics, immunogenicity, and exposure-response relationship of ustekinumab in patients with Crohn’s disease: Results from the week 16 interim analysis of the STARDUST study [UEG Week abstract OP042]. United European Gastroenterol J. 2020;8(Suppl):32.
-
Papamichael K, Cheifetz AS, Melmed GY, et al. Appropriate Therapeutic Drug Monitoring of Biologic Agents for Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2019;17(9):1655–68.
-
Singh S, Dulai PS, Vande Casteele N, et al. Systematic review with meta-analysis: association between vedolizumab trough concentration and clinical outcomes in patients with inflammatory bowel diseases. Aliment Pharmacol Ther. 2019;50(8):848–57.
-
Bressler B, Marshall JK, Bernstein CN, et al. Clinical practice guidelines for the medical management of nonhospitalized ulcerative colitis: the Toronto consensus. Gastroenterology. 2015;148(5):1035–58.
-
Singh S, Murad MH, Fumery M, et al. First- and Second-Line Pharmacotherapies for Patients With Moderate to Severely Active Ulcerative Colitis: An Updated Network Meta-Analysis. Clin Gastroenterol Hepatol. 2020;18:2179–91.
-
Xu Y, Hu C, Chen, et al. Population Pharmacokinetics and Exposure-Response Modeling Analyses of Ustekinumab in Adults With Moderately to Severely Active Ulcerative Colitis. J Clin Pharmacol. 2020;60(7):889–902.
-
Alsoud D, Vermeire S, Verstockt B. Monitoring vedolizumab and ustekinumab drug levels in patients with inflammatory bowel disease: hype or hope? Curr Opin Pharmacol. 2020;55:17–30.
-
Ma C, Battat R, Jairath V, et al. Advances in Therapeutic Drug Monitoring for Small-Molecule and Biologic Therapies in Inflammatory Bowel Disease. Curr Treat Options Gastroenterol. 2019;17(1):127–45.
-
de Almeida Martins C, Moss AC, Sobrado CW, et al. Practical Aspects of Proactive TDM for Anti-TNF Agents in IBD: Defining Time Points and Thresholds to Target. Crohn’s & Colitis. 2019;1(3):1–7.
-
Verstockt B, Moors G, Bian S, et al. Influence of early adalimumab serum levels on immunogenicity and long-term outcome of anti-TNF naive Crohn’s disease patients: the usefulness of rapid testing. Aliment Pharmacol Ther. 2018;48(7):731–9.
-
Feuerstein JD, Nguyen GC, Kupfer SS, et al. American Gastroenterological Association Institute Guideline on Therapeutic Drug Monitoring in Inflammatory Bowel Disease. Gastroenterology. 2017;153:827–34.
-
Turner D, Walsh CM, Steinhart AH, et al. Response to corticosteroids in severe ulcerative colitis: a systematic review of the literature and a meta-regression. Clin Gastroenterol Hepatol. 2007;5(1):103–10.
-
Herfarth HH, Rogler G, Higgins PD. Pushing the pedal to the metal: should we accelerate infliximab therapy for patients with severe ulcerative colitis? Clin Gastroenterol Hepatol. 2015;13(2):336–8.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC AGAF, Director, Division of Gastroenterology, Professor, Department of Medicine, McMaster University, Hamilton, ON
Contributing Author
Niels Vande Casteele, PharmD PhD, Assistant Professor, Department of Medicine, School of Medicine, Affiliate Faculty, Skaggs School of Pharmacy & Pharmaceutical Sciences, University of California San Diego, La Jolla, California USA
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Anne M. Griffiths, MC FRCPC, University of Toronto, Toronto, ON
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
IBD Dialogue 2021·Volume 17 is made possible by unrestricted educational grants from…
![]()
![]()




Published by Catrile & Associates Ltd., 167 Floyd Avenue, Toronto, ON M4J 2H9
(c) Catrile & Associates Ltd., 2021. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.